Teva and MedinCell Announce FDA Acceptance of New Drug Application for TV-46000/mdc-IRM as a Treatment for Patients with Schizophrenia | Business Wire

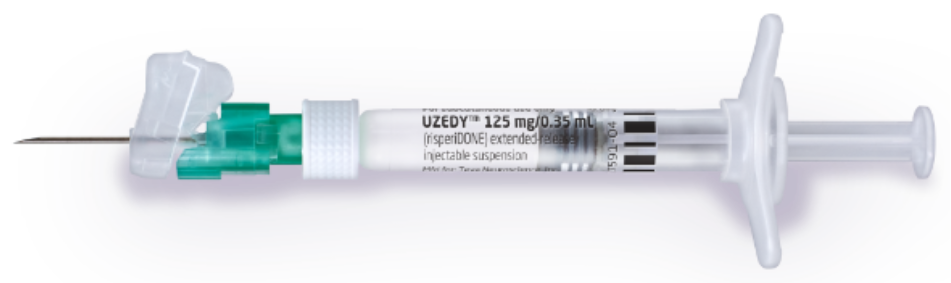
FDA Approves Uzedy (risperidone) Extended-Release Injectable Suspension for the Treatment of Schizophrenia in Adults

Teva and MedinCell Announce FDA Approval of UZEDY™ (risperidone) Extended-Release Injectable Suspension, a Long-Acting Subcutaneous Atypical Antipsychotic Injection, for the Treatment of Schizophrenia in Adults

Risperidone Teva 25 mg, proszek i rozpuszczalnik do sporządzania zawiesiny do wstrzykiwań o przedłużonym uwalnianiu, 1 zestaw | Allecco.pl