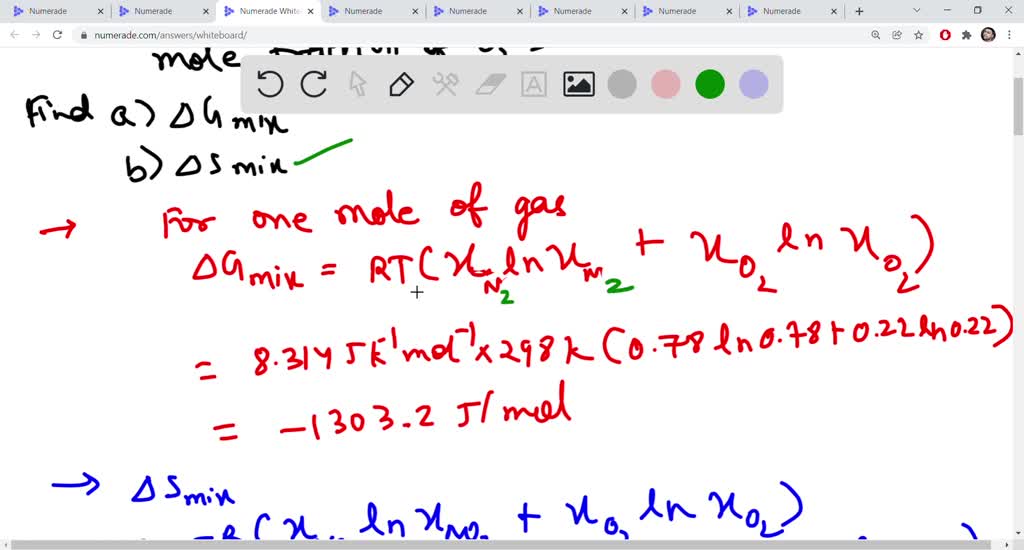
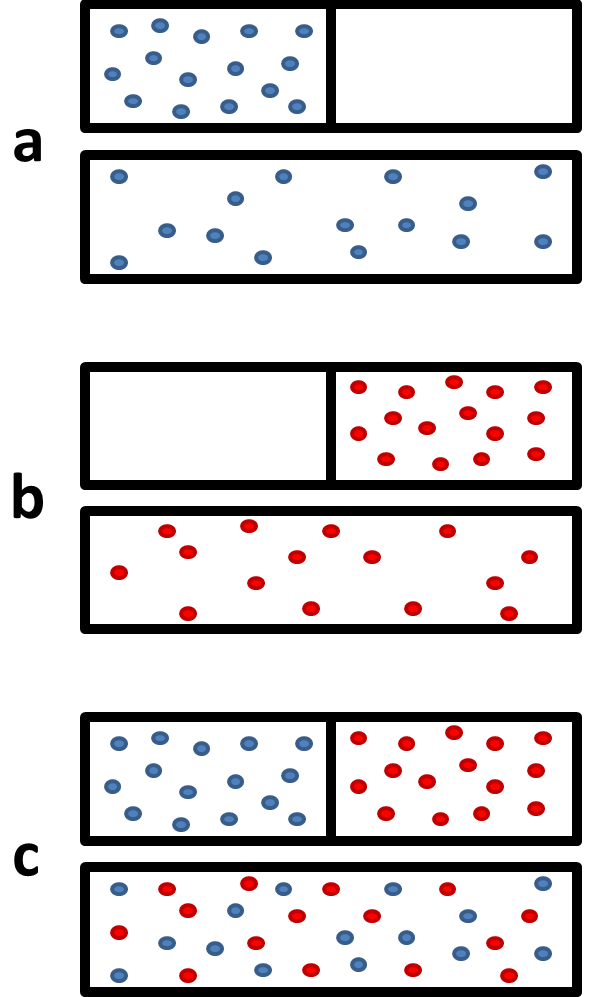
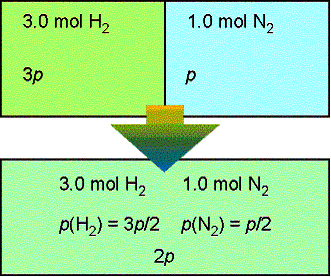
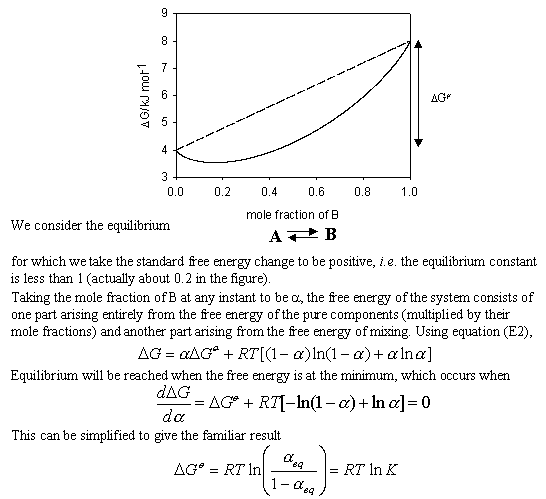Lecture 18Multicomponent Phase Equilibrium1 Theories of Solution The Gibbs energy of mixing is given by: And the chemical potential is: For ideal gases, - ppt download

Gibb's free energy of mixing plus Gibb's excess energy vs. water mole... | Download Scientific Diagram

SOLVED: 10.1 From Flory-Huggins theory the partial molar Gibbs free energy of 'mixing AG, with respect to the solvent for the formation of a polymer solution is given by AG, = 4-4" =