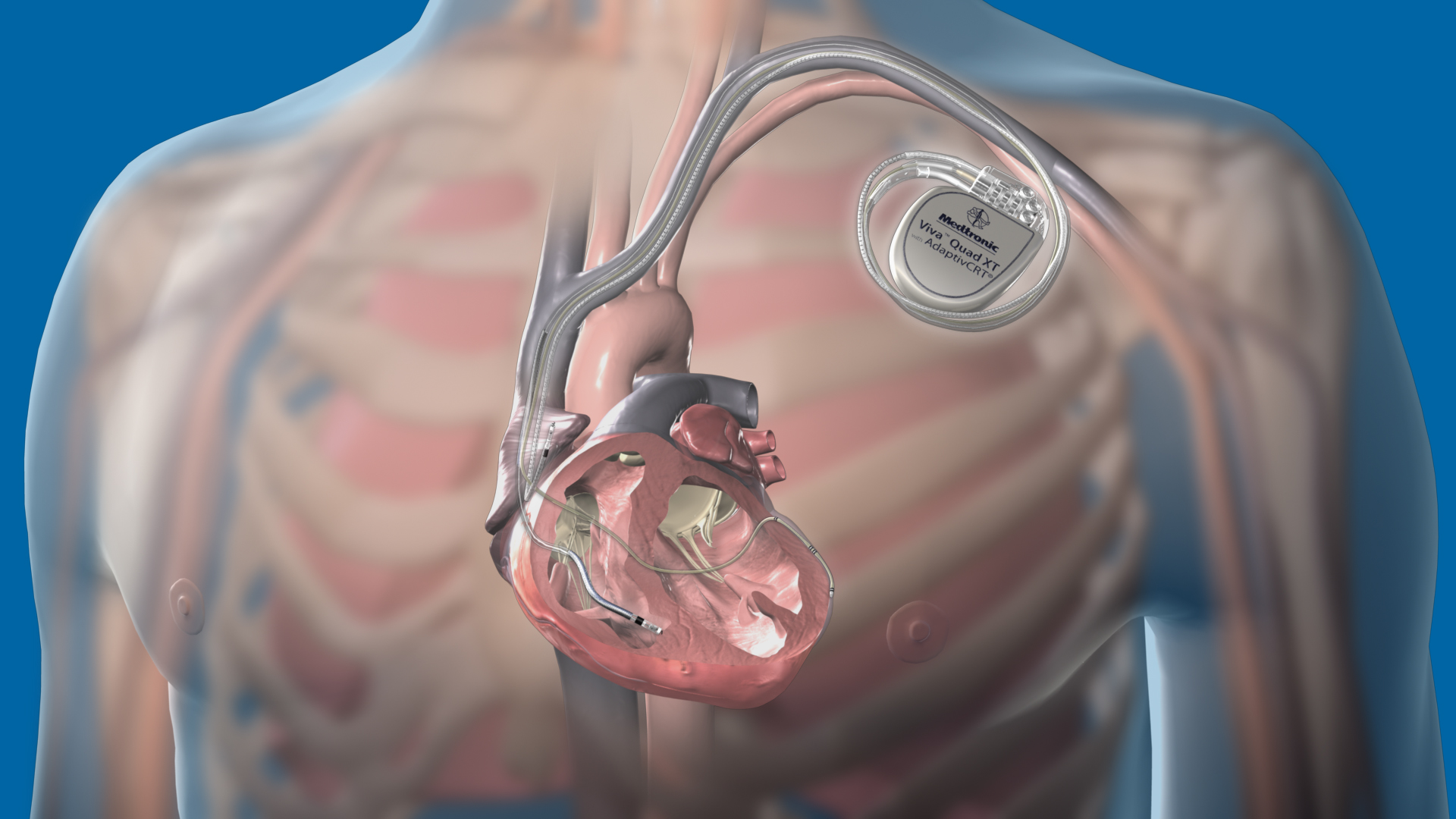
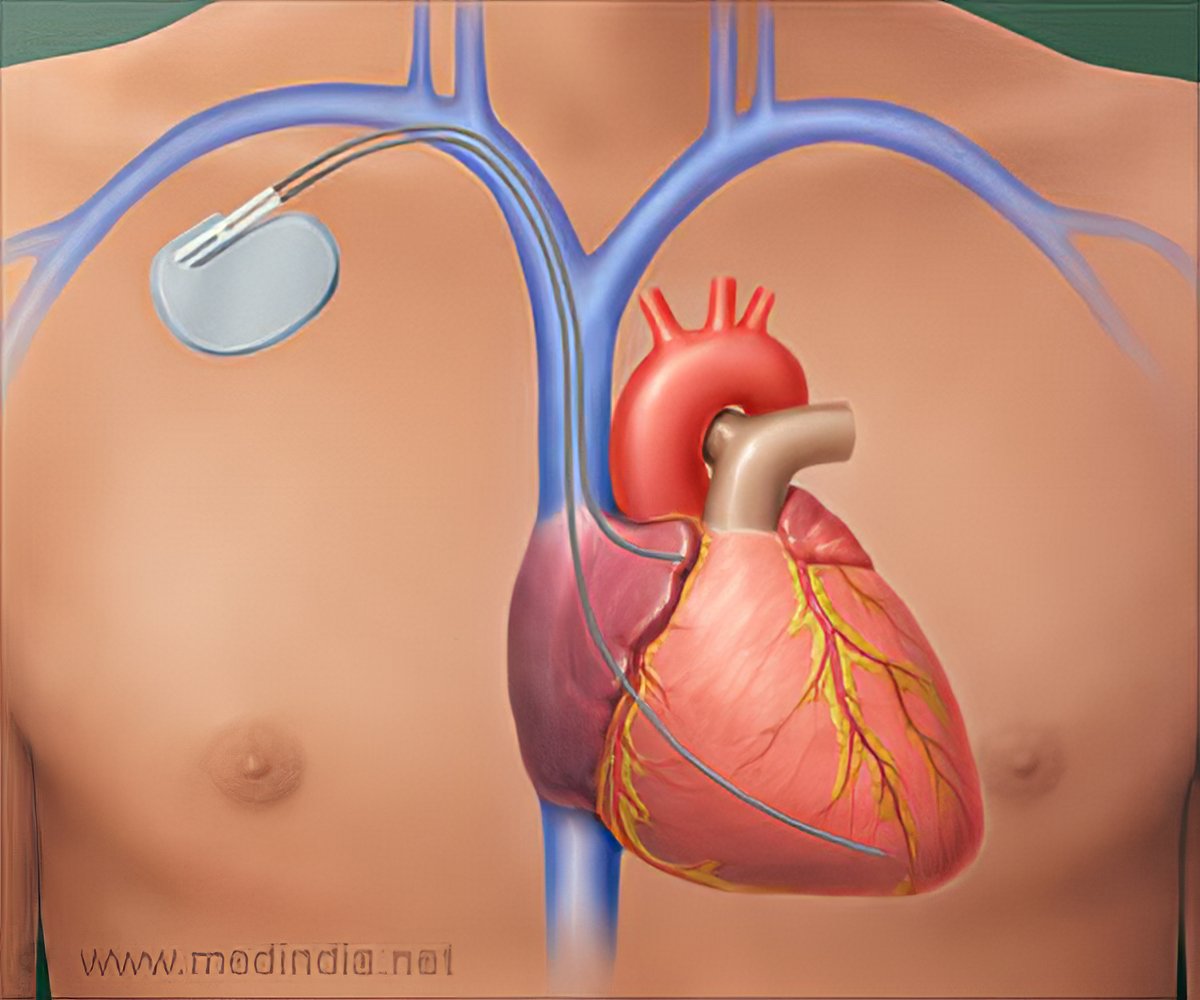
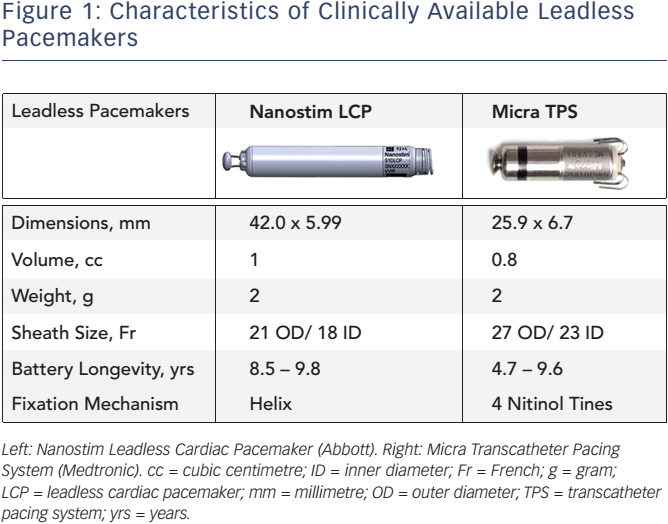
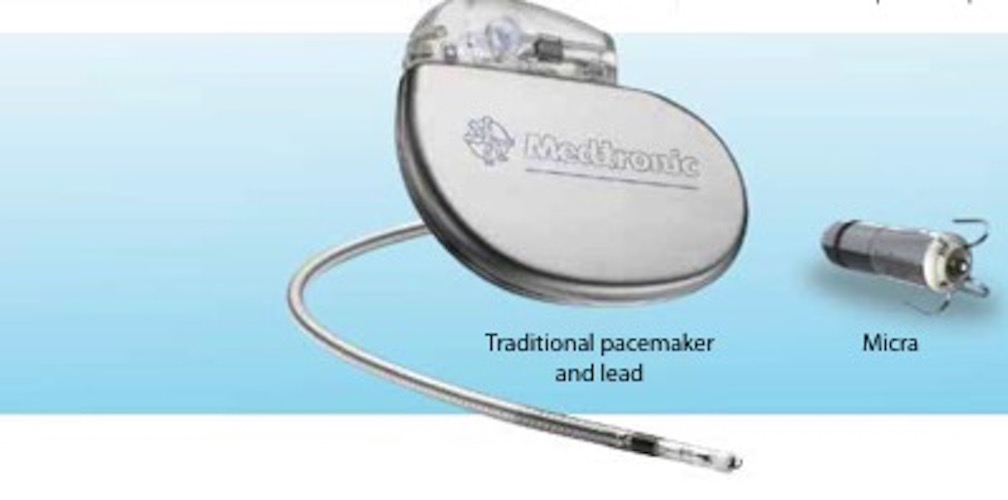
Perioperative Interrogation of Medtronic Cardiovascular Implantable Electronic Devices: A Guide for Anesthesiologists

The extravascular implantable cardioverter‐defibrillator: The pivotal study plan - Crozier - 2021 - Journal of Cardiovascular Electrophysiology - Wiley Online Library

Medtronic's Extravascular ICD Hits Targets In Trial; Will Soon Compete With Boston Scientific's S-ICD :: Medtech Insight

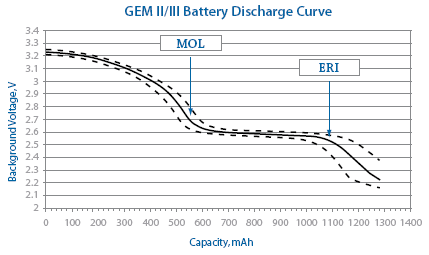
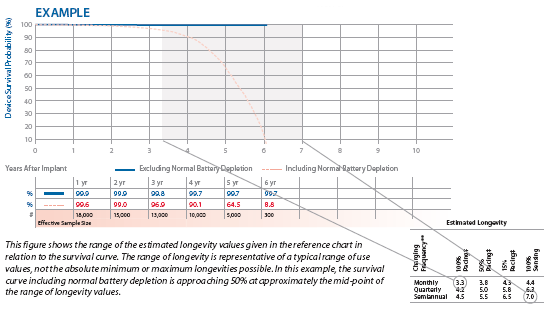
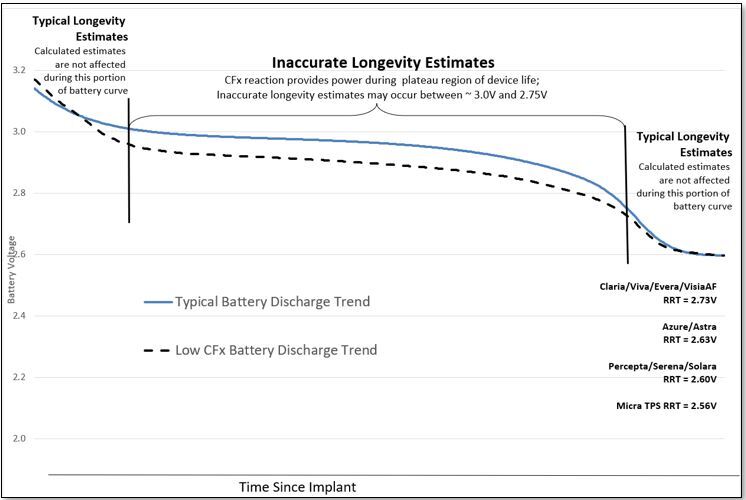
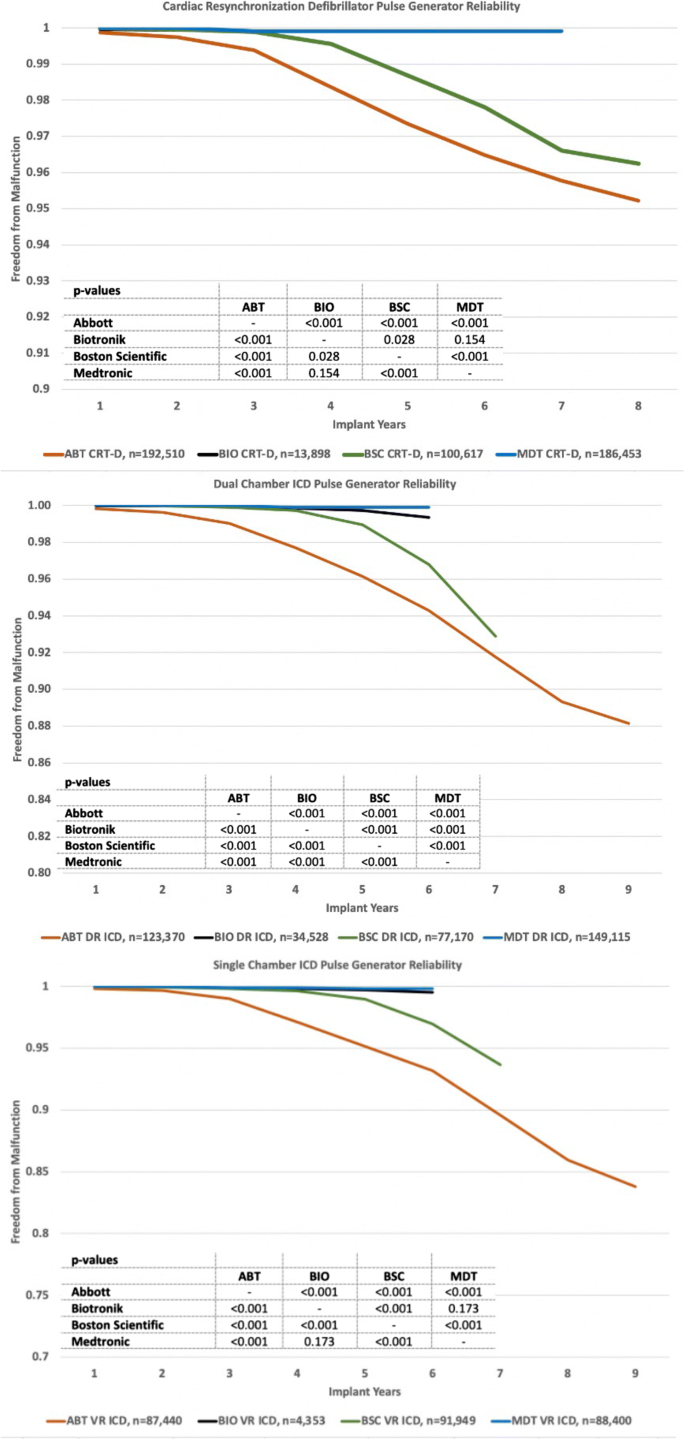
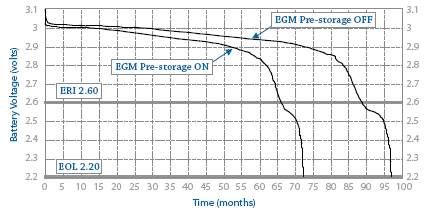
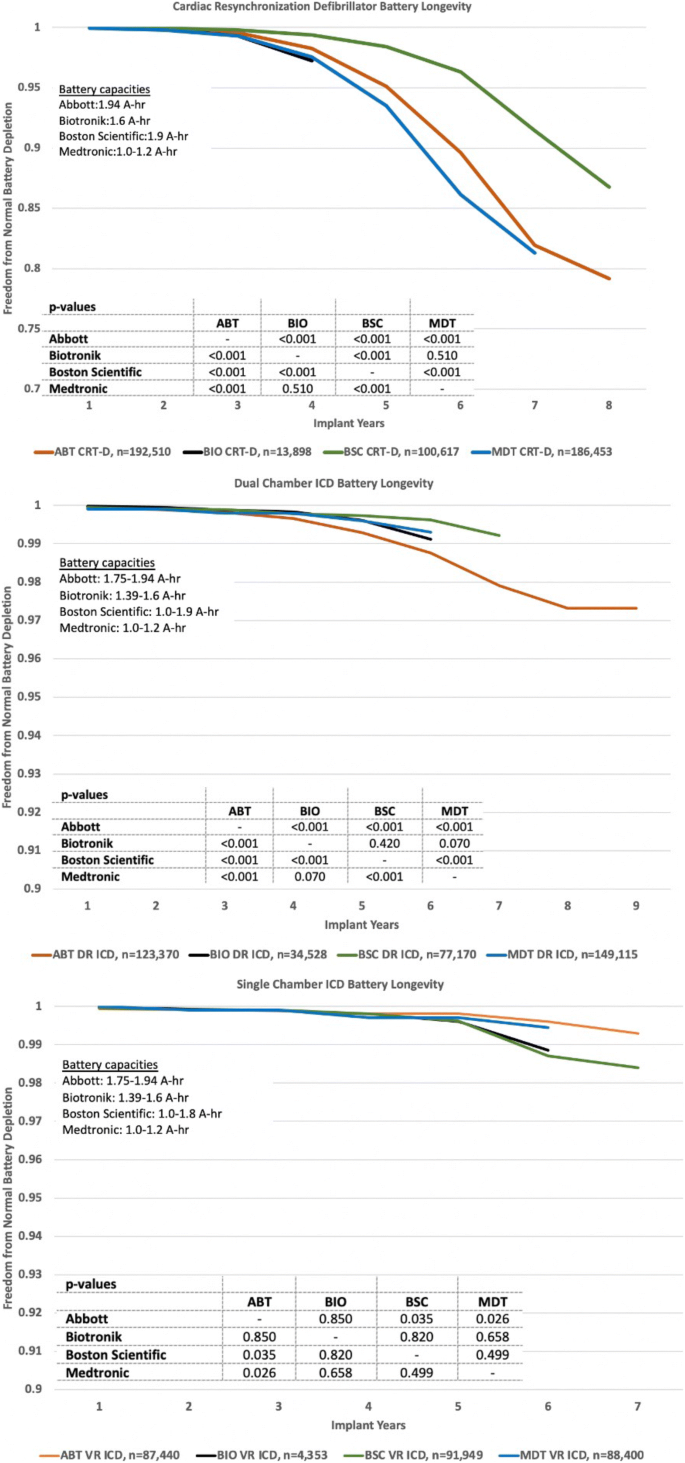
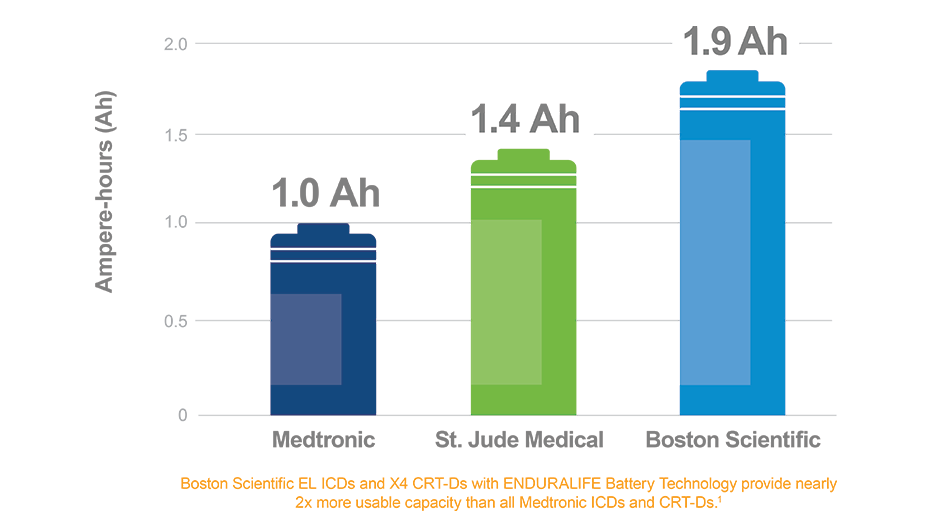
Predicted longevity of contemporary cardiac implantable electronic devices: A call for industry-wide “standardized” reporting - ScienceDirect