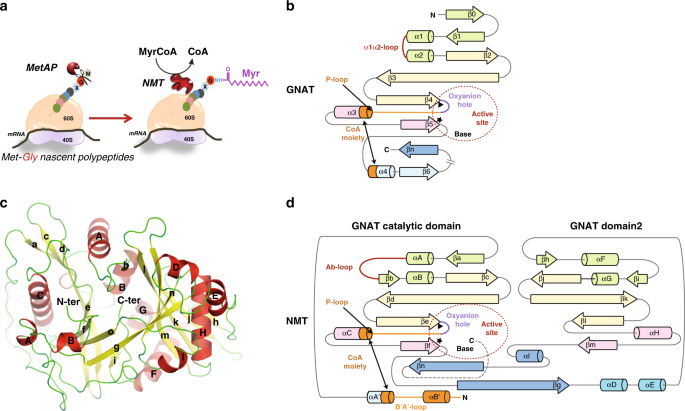
High-resolution snapshots of human N-myristoyltransferase in action illuminate a mechanism promoting N-terminal Lys and Gly myristoylation | Nature Communications

Exploiting phage receptor binding proteins to enable endolysins to kill Gram-negative bacteria | Scientific Reports
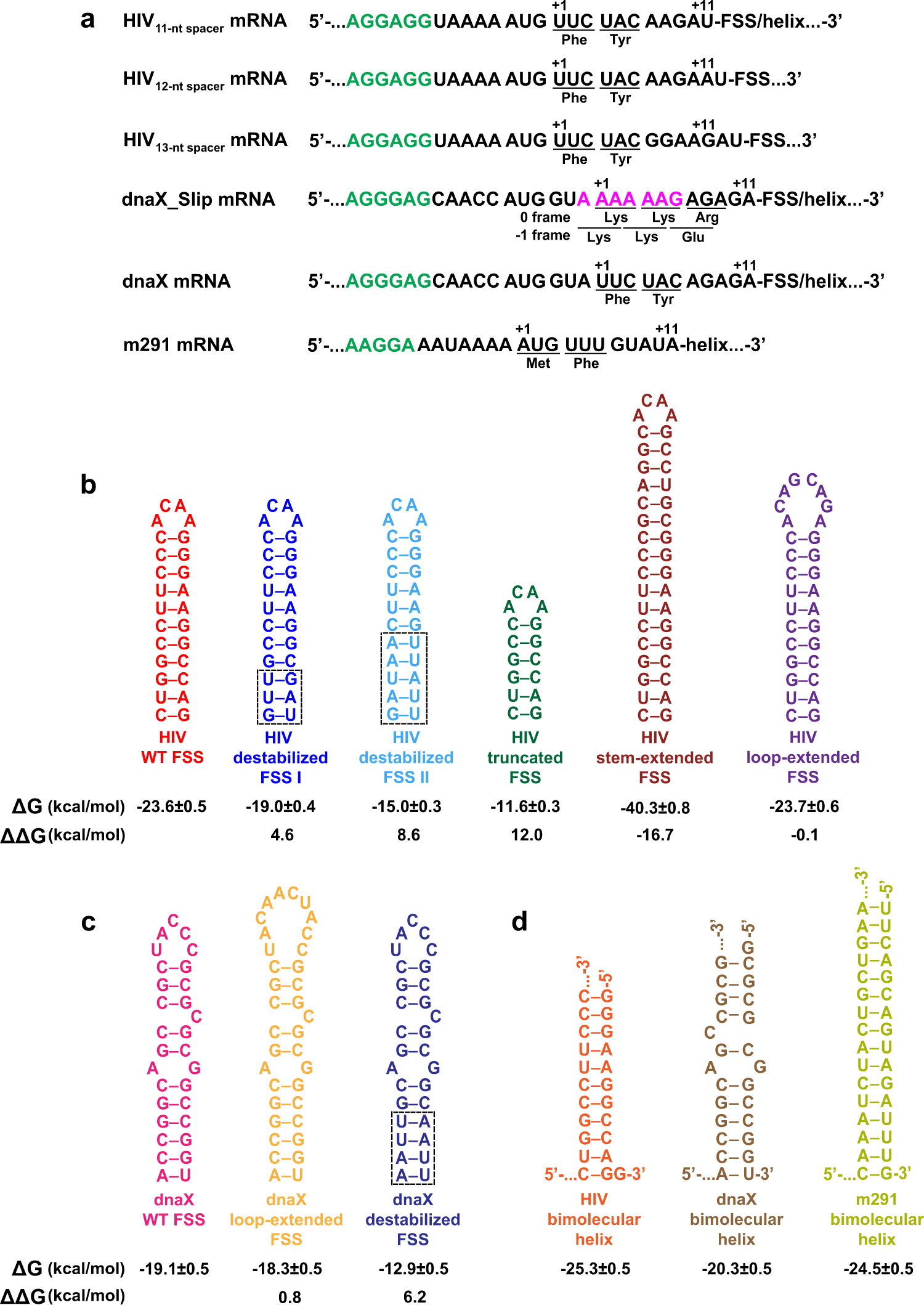
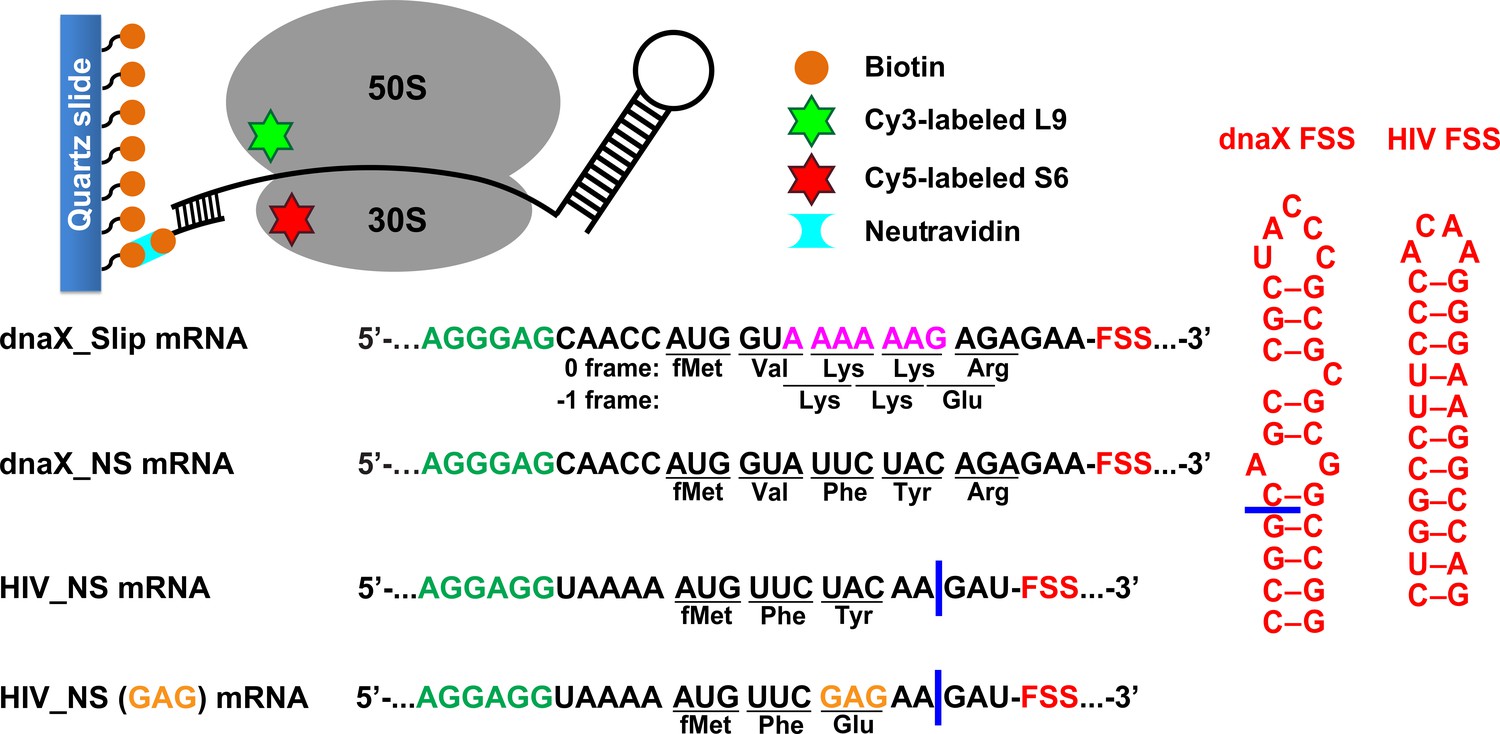
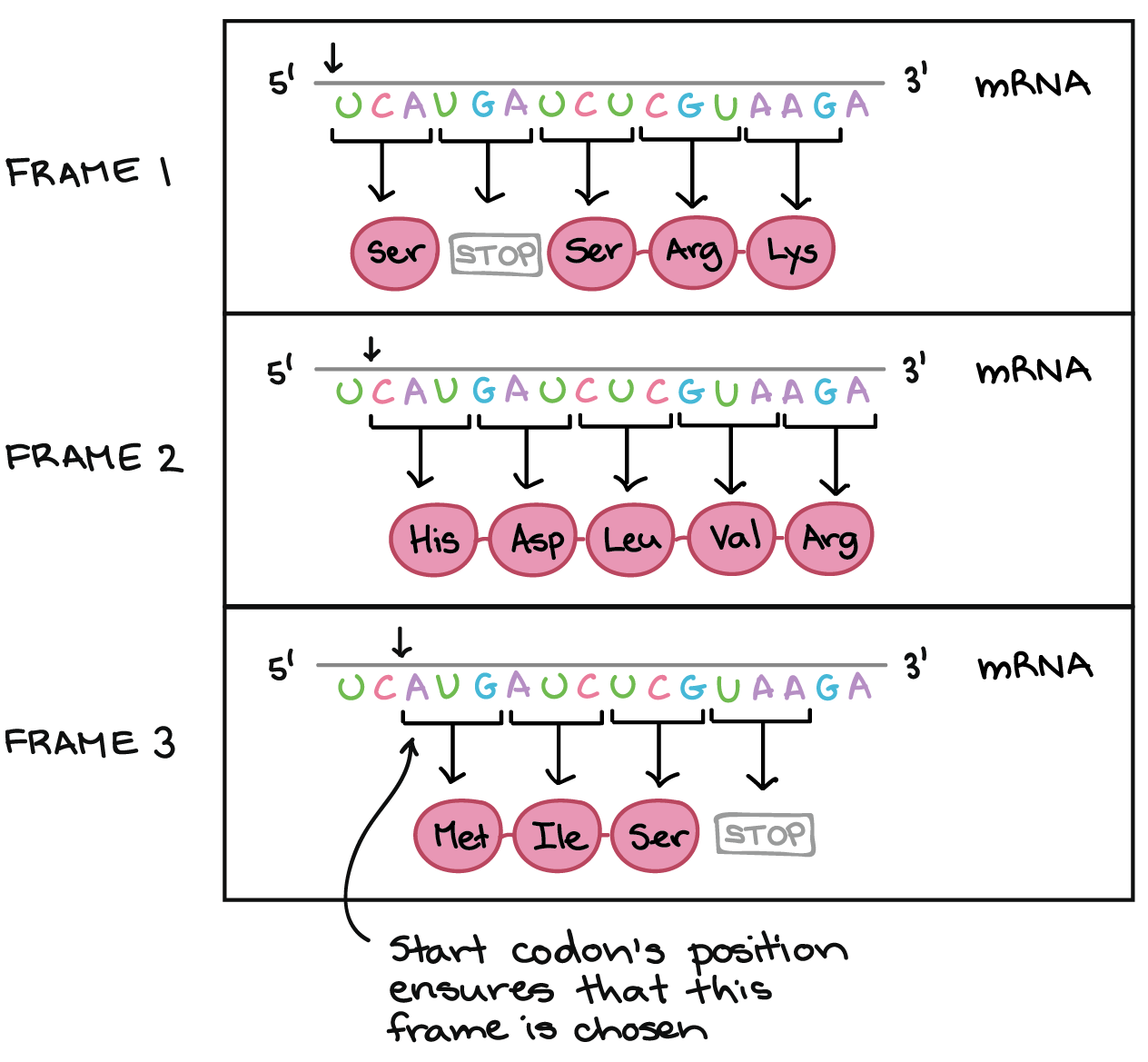
Specific length and structure rather than high thermodynamic stability enable regulatory mRNA stem-loops to pause translation | Nature Communications
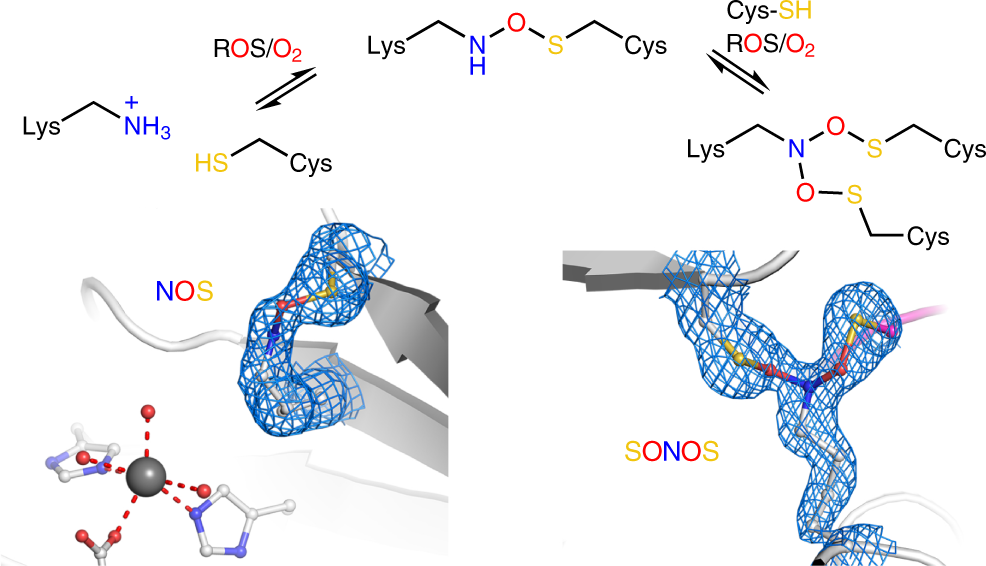
Widespread occurrence of covalent lysine–cysteine redox switches in proteins | Nature Chemical Biology
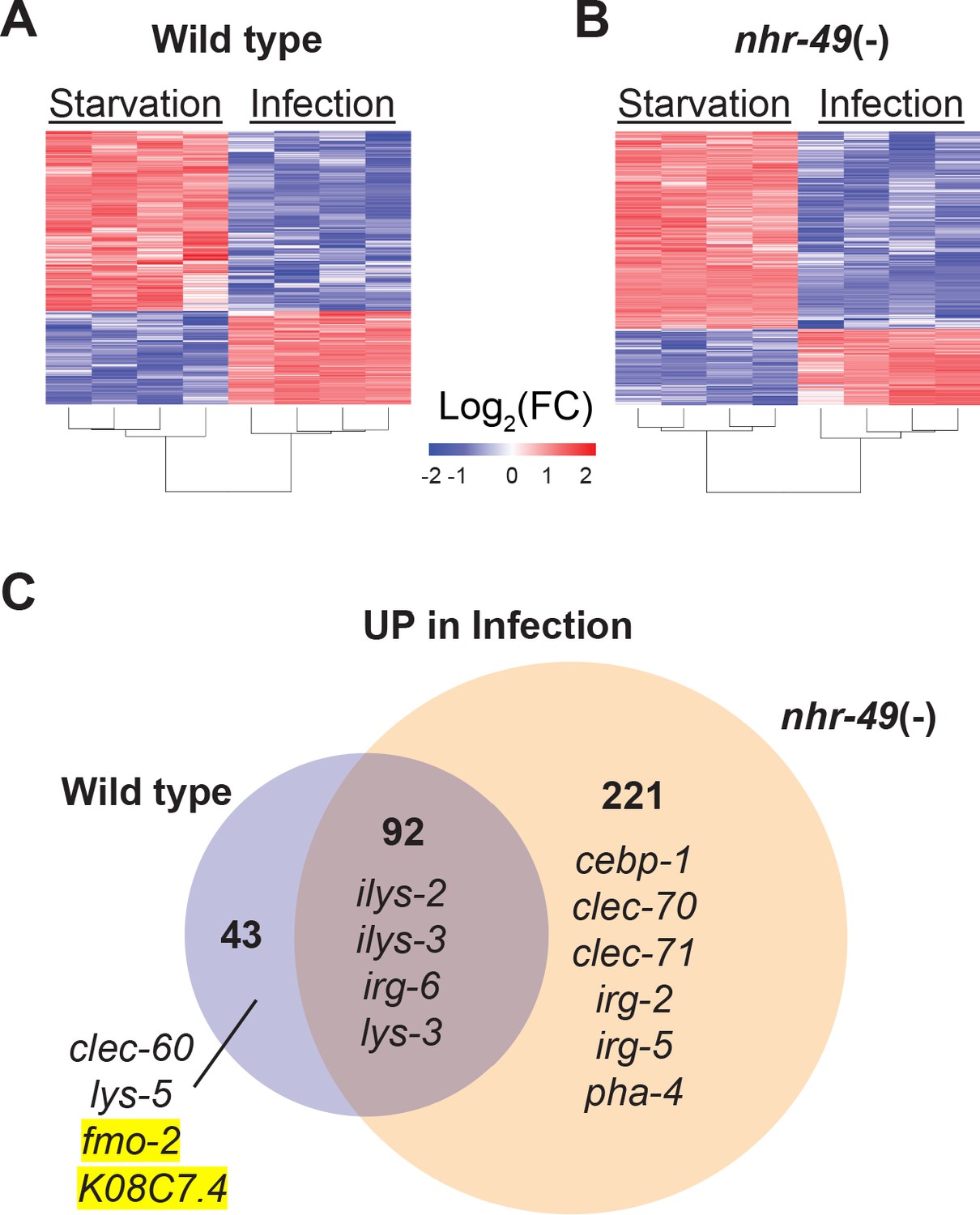
NHR-49/PPAR-α and HLH-30/TFEB cooperate for C. elegans host defense via a flavin-containing monooxygenase | eLife

Reversible Dual-Covalent Molecular Locking of the 14-3-3/ERRγ Protein–Protein Interaction as a Molecular Glue Drug Discovery Approach | Journal of the American Chemical Society