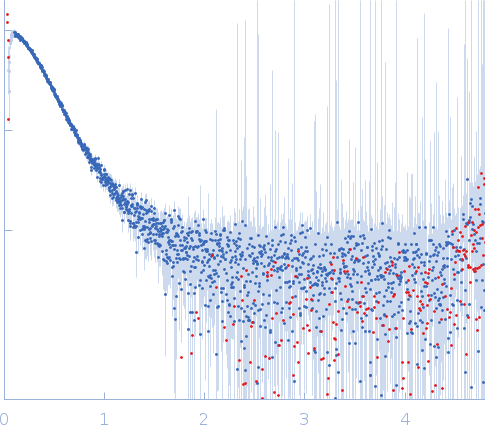
Structures of designed armadillo repeat proteins binding to peptides fused to globular domains - Hansen - 2017 - Protein Science - Wiley Online Library

An Armadillo motif in Ufd3 interacts with Cdc48 and is involved in ubiquitin homeostasis and protein degradation | PNAS

RCSB PDB - 2BAP: Crystal structure of the N-terminal mDia1 Armadillo Repeat Region and Dimerisation Domain in complex with the mDia1 autoregulatory domain (DAD)

Biomolecules | Free Full-Text | Folding and Stability of Ankyrin Repeats Control Biological Protein Function
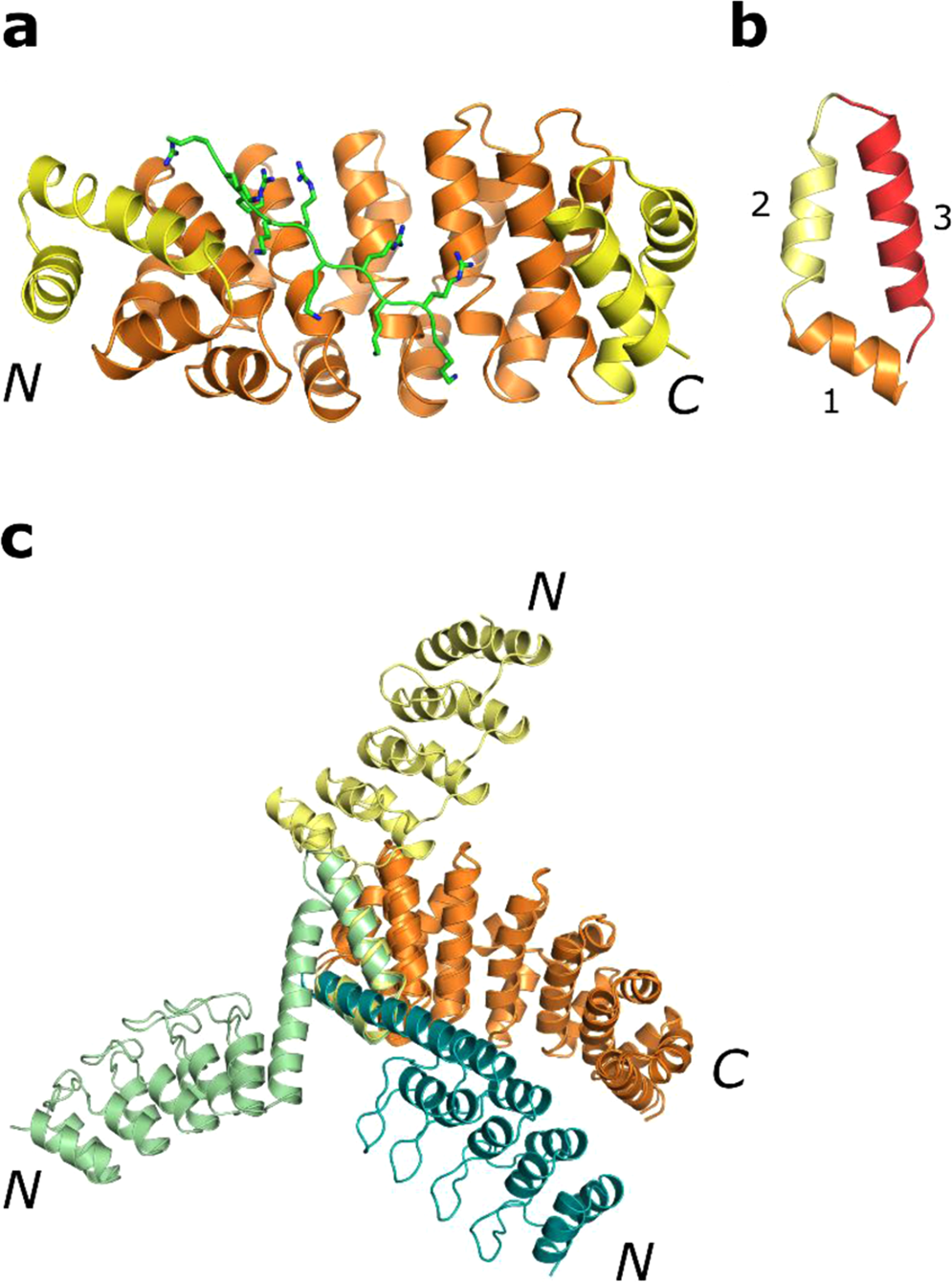
Rigid fusions of designed helical repeat binding proteins efficiently protect a binding surface from crystal contacts | Scientific Reports
4PLQ: Crystal Structures Of Designed Armadillo Repeat Proteins: Implications Of Construct Design And Crystallization Conditions On Overall Structure

Designed Armadillo Repeat Proteins: Library Generation, Characterization and Selection of Peptide Binders with High Specificity - ScienceDirect
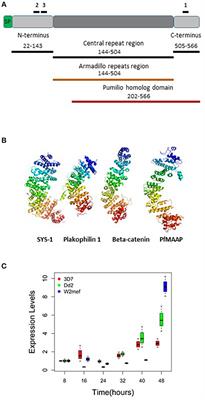
Plasmodium falciparum Merozoite Associated Armadillo Protein (PfMAAP) Is Apically Localized in Free Merozoites and Antibodies Are Associated With Reduced Risk of Malaria - Frontiers
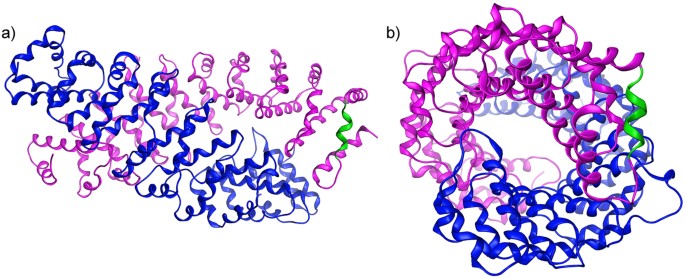
A motif within the armadillo repeat of Parkinson's-linked LRRK2 interacts with FADD to hijack the extrinsic death pathway | Scientific Reports

Biomolecules | Free Full-Text | Parallel and Sequential Pathways of Molecular Recognition of a Tandem-Repeat Protein and Its Intrinsically Disordered Binding Partner



![PDF] Armadillo-repeat protein functions: questions for little creatures. | Semantic Scholar PDF] Armadillo-repeat protein functions: questions for little creatures. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0884c0654d47ef1c982e352cd3ee885c161a4534/6-Figure3-1.png)





![PDF] Armadillo repeat proteins: beyond the animal kingdom. | Semantic Scholar PDF] Armadillo repeat proteins: beyond the animal kingdom. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a56e4629ab2b920e867b0b6ac5a556e2d5d08ff1/8-Figure1-1.png)