Cephalon sales rep tells court Actiq, Fentora opioids were not viable sellers in W.Va. | West Virginia Record

Israeli drug company Teva reaches $4.3 billion settlement in US opioid lawsuits | The Times of Israel

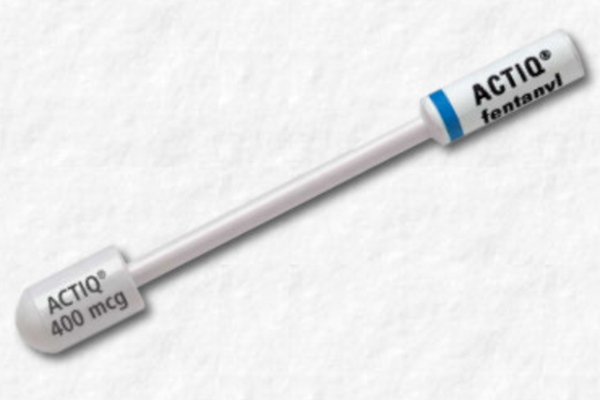
These highlights do not include all the information needed to use ACTIQ safely and effectively. See full prescribing information for ACTIQ. ACTIQ® (fentanyl citrate) oral transmucosal lozenge, CII Initial U.S. Approval: 1968